 
The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". 1.Very close relative to the size of the molecules, 2.Close relative to the size of the molecules, 3.Fixed relative to the size of the molecules, 4.Small relative to the size of the molecules, 5. This cookie is set by GDPR Cookie Consent plugin. In a gas, the distance between the particles is. The cookie is used to store the user consent for the cookies in the category "Other. This cookie is set by GDPR Cookie Consent plugin. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. The molecules in a gas are known to be significantly far from each other. Gas is one of the states of matter, the other two being liquid and solid, which molecules are randomly distributed in space. How are the molecules in a gas distributed? The distance between particles in a gas is much larger than the distance between particles in a solid or a liquid. In gases the atoms and molecules are free to move independently, colliding frequently. Gases can be spread apart or be squezed together as they fill a container. How are gases different from solid and liquid? The probability to find a particle at the distance from the origin between and is ,… An NN particle at distance means exactly one of the particles resides at that distance while the rest particles are at larger distances, i.e., they are somewhere outside the sphere with radius. Plot of variation of ion pair current against applied voltage for a wire cylinder gaseous radiation detector. What does it mean when a particle is at a distance? Are gases are made of particles that are far apart compared with those of liquids and solids? Usually yes, but the distance between the particles depends on pressure. Which is larger the distance between particles in a gas or solid? The average distance between the particles would be the distance between the centerpoints of two neighboring balls each with volume V/N. How do you calculate the average distance between gas molecules? In which state are the distances between the particles greatest?Įxpert Answers In a gas the distance between its molecules (or atoms) is the highest from all three states of matter because the interaction forces between them are the weakest. The gas particles have big distances between them. The particles in a liquid usually are still touching but there are some spaces between them. 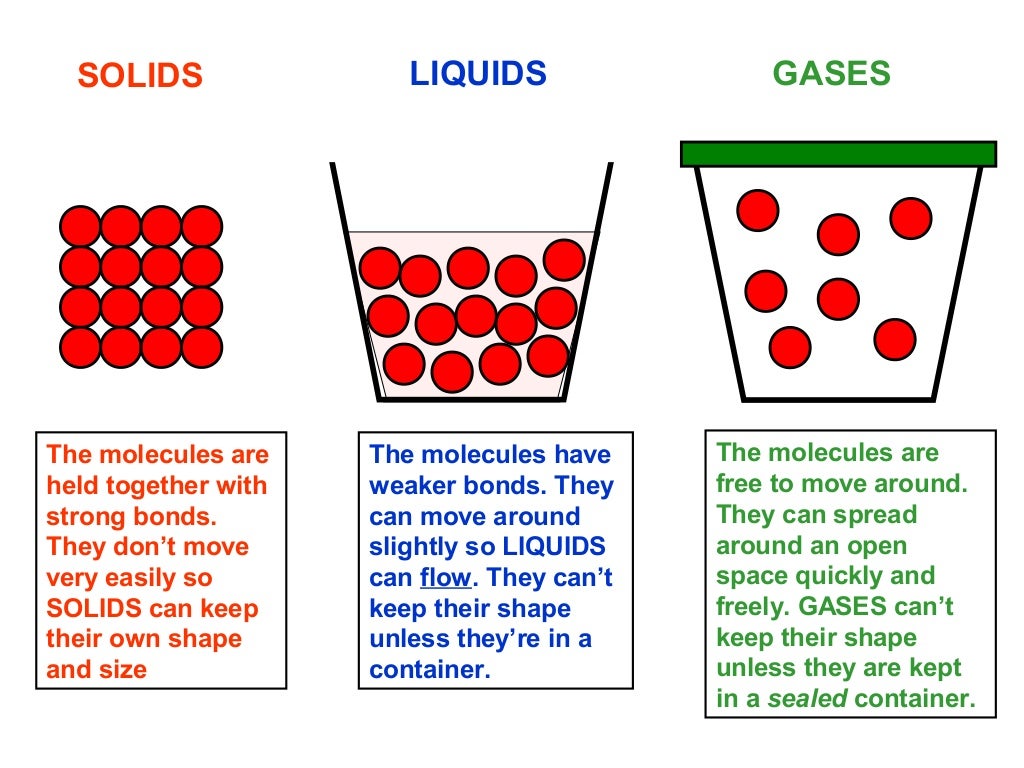
The particles in the solid are touching with very little space between them. How is the spacing between the particles different in solid liquid and gas? Between particles, there’s just empty space. Within a gas, the particles of a gas are relatively far apart compared with the distance between particles in a liquid or a solid. Many are electrostatic in nature and therefore depend on the distance between particles. There are a number of interactions that change the energy of molecules and ensembles of molecules. The particles in a gas are considered to be small, hard spheres with an insignificant volume. The cause of non-ideal behavior is intermolecular interactions. What is found in between the particles of a gas? The molecules in a gas are very far apart so they can be squeezed together into a smaller space. The average kinetic energy of gas particles is dependent on the temperature of the gas. The size of gas particles is tiny compared to the distances that separate them and the volume of the container. Gas particles are separated by large distances. 6 What does it mean when a particle is at a distance?Īre gas particles separated by large distances?.5 Which is larger the distance between particles in a gas or solid?.4 How do you calculate the average distance between gas molecules?.3 What is found in between the particles of a gas?.1 Are gas particles separated by large distances?. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
